1 Fulminate is the anion CN+O or any of its salts. It is an isomer of isocyanic acid ( HNCO) and of its elusive tautomer, cyanic acid ( HOCN ), and also of isofulminic acid ( HON+C ). Yes, just switch the single and double bonds for the above two structures (with the oxygens and their lone electrons). Fulminic acid is an acid with the formula HCNO, more specifically HCN+O. 
Write the Lewis structure and chemical formula of the compound with a molar mass of about 70 g/mol that contains 19.7% nitrogen and 80.3% fluorine by mass, and determine the formal charge of the atoms in this compound.In the left resonance structure, all the atoms have zero formal charge, while on the right structure, the nitrogen has a +1 formal charge, and the oxygen with the single bond has a -1 formal charge.Ĭan you draw two additional resonance structures for the above molecules?.Write Lewis structures for each of the four compounds and determine the formal charge of the iodine atom in each molecule: Iodine forms a series of fluorides (listed below).Is the actual structure consistent with the formal charges? Draw the structure of hydroxylamine, H 3NO, and assign formal charges look up the structure.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in sulfur dioxide: OSO or SOO?.Step 2: Calculate the formal charge of the compound using the Lewis Dot Structure in step 1 and. Subtract the number of non-bonding electrons (usually in lone pairs). Step 1: Draw the Lewis Dot Structure for the compound given in order to calculate the formal charge. 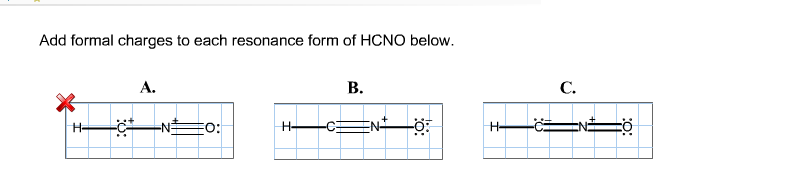
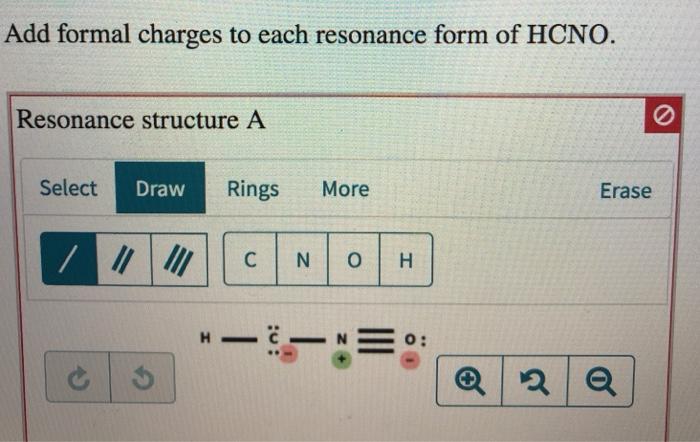
We use the following equation to calculate formal charge on an atom in Lewis structure. Formal charge on each atom is due to difference in valence electrons in an isolated atom and number of electrons assigned to that atom in a Lewis structure. Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in hypochlorous acid: HOCl or OClH? Calculating formal charge Count the number of valence electrons of the neutral atom. Each atom in a Lewis structure for a molecule or ion will have formal charge.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in nitrosyl chloride: ClNO or ClON?.Determine the formal charge on each atom in each of the resonance structures: Add formal charges to each resonance form of HCNO. Draw all possible resonance structures for each of the compounds below.Atom Structure 1 Structure 2 Structure 3 S N b) Which of the three structures listed above is the best Lewis structure for this. Structure 1 Structure 2 Structure 3 :S EN: S. Thus, we calculate formal charge as follows: a) For the three Lewis structures shown below, determine the formal charge of each of the three atoms (S, C, and N). Another way of saying this is that formal charge results when we take the number of valence electrons of a neutral atom, subtract the nonbonding electrons, and then subtract the number of bonds connected to that atom in the Lewis structure. The formula for the formal charge on any atom in the Lewis structure is Formal charge number of valence electrons - number of bonds made by the atom. The formal charge of an atom in a molecule is the hypothetical charge the atom would have if we could redistribute the electrons in the bonds evenly between the atoms. We can use the concept of formal charges to help us predict the most appropriate Lewis structure when more than one is reasonable. As we have seen, however, in some cases, there is seemingly more than one valid structure for a molecule. In order to calculate the formal charges for HCN well use the equation:Formal charge of valence electrons - nonbonding val electrons - bonding elec. Justify your choice based on formal charges. H-CN-O: H-CNO (a) Explain why the diagram on the left is the better representation for the bonding in fulminic acid. In the previous section, we discussed how to write Lewis structures for molecules and polyatomic ions. Two possible Lewis electron-dot diagrams for fulminic acid, HCNO, are shown below. Explain the concept of resonance and draw Lewis structures representing resonance forms for a given molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
